Investigations into Tumor Dormancy
Tumor dormancy — a state of tumor development characterized by halted growth — is highly prevalent in both cancer patients and normal populations. The state of tumor dormancy may occur throughout various stages of tumor development. Clinical and experimental observations suggest micro-metastasis in distant organs may be dormant for extended periods, as may minimal residual disease left after surgical removal of the tumor mass or other treatment of primary tumors. For example, often patients believed to have a good prognosis post-therapy, nonetheless develop metastases, in some cases many years after apparently successful treatment of their primary cancer. The concept of grossly uneven rates of tumor growth extend to larger tumors as well. Mammographies are revealing new tumor lesions which, based on rates of growth calculated post-detection, should not have been missed on the previous mammogram unless the previous growth rates of these tumors were much faster and they subsequently entered a state of much slower growth or near-dormancy. These punctuated periods of attenuated growth throughout tumor development lead to many questions, such as how best to assign risk of late recurrence, how long to monitor patients, what mechanisms regulate tumor dormancy, and which mechanisms enable tumors to advance from the dormant state and renew tumor mass expansion.
Despite the high prevalence of dormant tumors, little is known and much remains to be learned about tumor dormancy. Both experimental studies, as well as computational and systems mathematical modeling done at our CCSB are shedding light on the biological and molecular mechanisms responsible. Kinetically, two mechanisms have been proposed. Single-cell dormancy has been attributed to cell cycle arrest, while dormancy of larger tumor masses has been attributed to balanced proliferation and cell death. Cell-intrinsic, population-intrinsic, and host interaction effects are potential causes. Among the latter, the immune system, angiogenesis, and niche ‘permissivity’ are recognized as potential bottlenecks to tumor progression. These may operate independently. Our group has recently developed a dormancy model in immunodeficient mice, pointing to angiogenesis as a likely controller of exit from dormancy in this case.
Angiogenesis, Dormancy, and the Timeline Paradigm for Tumor Development
Interestingly, the “angiogenic switch” and a “shift in the angiogenic balance” have been used interchangeably to describe the angiogenesis-dependent transition from dormancy to aggressive growth (see e.g., Bouck N, Adv Cancer Res 69: 135-74, 1996). Each, however, really carries a different implication. A “switch” suggests a potentially irreversible transition from a non-angiogenic to an angiogenic state, and has been used often in reference to the acquisition of vasculature by a small, avascular tumor which allows it to grow beyond the oxygen diffusion distance. This has been one major focus of our dormancy studies. Another of our focuses is centering on the angiogenic “balance” — an active dynamic between angiogenic and non-angiogenic states that may shift in either direction depending on ambient conditions. There is reason to believe such a dynamic remains operative even in larger, vascularized tumors. Indeed, the concept of antiangiogenic therapy relies, in essence, on the ability to revert clinical angiogenic tumors back to a non-growing state by re-establishing a limiting angiogenic balance or even an imbalance in favor of regression. This notion raises exciting questions about the control of tumor growth throughout progression. In particular, it points to a need to augment models — e.g., the Two-Stage Clonal Expansion (TSCE) Model (see Fig. 1 below) — which take various cell-level carcinogenesis events into account, but ultimately equate cancer disease risk to the occurrence of the first cancer cell. As shown in Fig. 2 below, the inevitability of cancer disease assumed by some models after the first cancer cell (“RISK=1” at left) may actually be far less than inevitable (“LOW RISK”), with the difference dictated by whether the tumors can breach subsequent angiogenic, invasive, and other host permissivity barriers to growth.
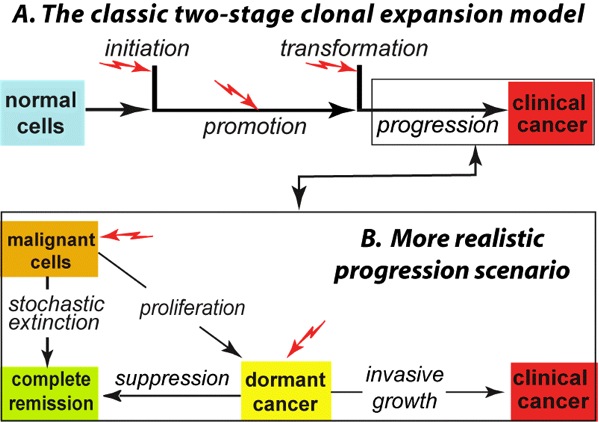
|
Fig. 1. Panel A depicts the venerable two-stage clonal expansion (TSCE) mathematical carcinogenesis model. Here: initiation is an alteration, such as a mutation, in a normal cell that makes it a stage-1 (that is, pre-malignant) cell; promotion is stochastic proliferation of pre-malignant cells; transformation is another alteration, generating a stage-2 (i.e., malignant) cell from the pool of pre-malignant cells; progression occurs during the time from the production of the first malignant cell to clinically observable cancer. It has often been modeled simply as a fixed lag time. Jagged red arrows indicate possible radiation influences. Single initiation or transformation steps are very brief compared to the promotion and progression steps, which may last many years. More sophisticated models in the literature involve several successive initiation steps. Panel B takes into account dormancy, which gives a more realistic mathematical model that strongly alters risk predictions. |
| Fig. 2. In light of the existence of progression-level bottlenecks to tumor growth, the assumption that the risk of cancer disease is certain once a cancer cell is created is in question. The likelihood a tumor will be halted at one of these bottlenecks correspondingly lowers the risk of final cancer disease assessed at the time of the creation of the first cancer cell (shown by the drop in the blue line from “RISK=1” to “LOW RISK”). | 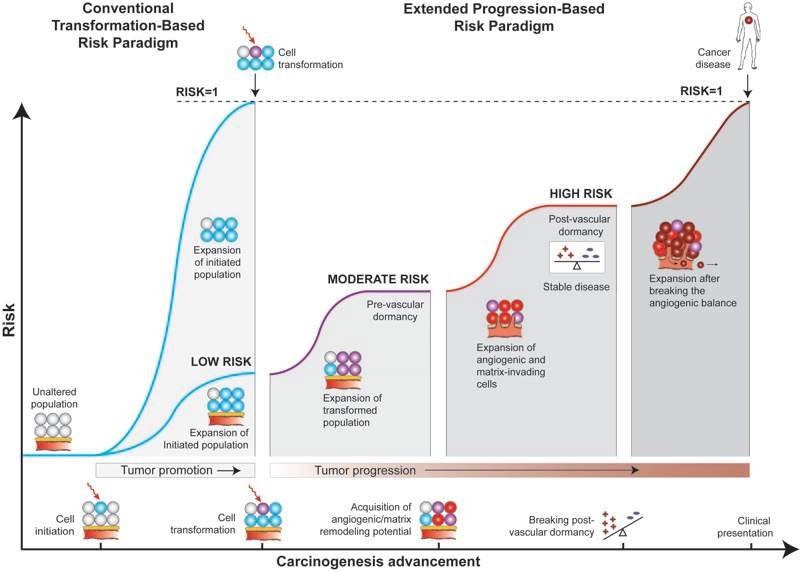 |
CCSB researchers have expanded on these studies experimentally and theoretically. Taking a broad approach, our group has utilized multi-disciplinary approaches to further explore tumor dormancy:
- Combined expertise in molecular and cellular biology to address the underlying mechanisms that lead to tumor dormancy;
- Used computational models to help dissect the dependencies on cell proliferation, cell crowding, migration and prevalence of tumor stem cells;
- Explored platelets biology in search of ultra-early biomarkers for presence of dormant tumors;
- Calculated mathematical predictions to investigate how dynamics in angiogenesis potential affect tumor growth and dormancy.
Resources
Publications:
The following articles on the topic of tumor dormancy have been published by researchers at CCSB (Click on title to go to
manuscript abstract):
- Beheshti A, Benzekry S, McDonald JT, Ma L, Peluso M, Hahnfeldt P, Hlatky L. Host age is a systemic regulator of gene expression
impacting cancer progression. Cancer Res. 2015 Mar
15;75(6):1134-43. Epub 2015 Mar 2. PMCID: PMC4397972
- Poleszczuk J, Hahnfeldt P, Enderling H. Evolution and phenotypic selection of cancer stem cells. PLoS
Comput Biol. 2015 Mar 5;11(3):e1004025. PMCID: PMC4351192 [Open Access]
- Shaked Y, McAllister S, Fainaru O, Almog N. Tumor dormancy and the angiogenic switch: possible implications of bone marrow-derived cells. Curr Pharm Des. 2014;20(30):4920-33. Epub 2013 Nov 25.
- Girdhani S, Lamont C, Peluso M, Sun M,
Hlatky L. 56Fe ion irradiation enhances angiogenesis and other
inter-cellular determinants of carcinogenesis risk. J Radiat Res. 2014 Mar 1;55 Suppl
1:i124-i126. PMCID: PMC3941549 [Open Access]
- Hlatky L, Hahnfeldt P. Beyond the cancer cell: progression-level determinants highlight the multi-scale nature of carcinogenesis risk. Cancer Res. 2014 Feb 1;(3):659-64. Epub 2013 Nov
22. [Open Access]
- Benzekry S, Gandolfi A, Hahnfeldt P. Global dormancy of metastases due to systemic inhibition of angiogenesis. PLoS One. 2014
Jan 21;9(1):e84249. eCollection 2014 Jan 21. PMCID: PMC3897365 [Open Access]
- Wilkie KP, Hahnfeldt P. Mathematical models of
immune-induced cancer dormancy and the emergence of immune
evasion. Interface Focus. 2013 Aug 6;3(4):20130010. Epub 2013 Jun
25. PMCID: PMC3915830 [Open Access]
- Almog N, Briggs C, Beheshti A, Ma L,
Wilkie KP, Rietman E, Hlatky
L. Transcriptional changes induced by the tumor
dormancy-associated microRNA-190. Transcription. 2013 Jul 1;4(4). PMCID: PMC3977918 [Open Access] COVER ARTICLE
- Wilkie KP, Hahnfeldt P. Tumor–immune dynamics regulated in the microenvironment inform the transient nature of immune-induced tumor dormancy. Cancer Res. 2013 Jun 15;73(12):3534-44. Epub 2013 Mar 27. PMCID: PMC3955200 [Open Access]
- Enderling H, Almog N, Hlatky L, editors. Systems Biology of Tumor Dormancy. New
York: Springer, 2013: 291p. In Series: Advances in Experimental Medicine and Biology, Vol. 734.
- Almog N. Genes and regulatory pathways involved in persistence of dormant micro-tumors. Adv Exp Med Biol. 2013;734:3-17.
- Hahnfeldt P. The host support niche as a control point for tumor dormancy: implications for tumor development and beyond. Adv Exp Med Biol. 2013;734:19-35.
- Enderling H. Cancer stem cells and tumor dormancy. Adv Exp Med Biol. 2013;734:55-71.
- Wilkie KP. A review of mathematical models of cancer-immune interactions in the context of tumor dormancy. Adv Exp Med Biol. 2013;734:201-34.
- Satchi-Fainaro R, Ferber S, Segal E, Ma L, Dixit N, Ijaz A, Hlatky L, Abdollahi A, Almog N. Prospective identification of glioblastoma cells generating dormant tumors. PLoS One. 2012;7(9):e44395. Epub 2012 Sep 6. PMCID: PMC3435314 [Open Access]
- Almog N, Ma L, Schwager C, Brinkmann BG, Beheshti A, Vajkoczy P, Folkman J, Hlatky L, Abdollahi A. Consensus micro RNAs governing the switch of dormant
tumors to the fast-growing angiogenic phenotype. PLoS One. 2012;7(8):e44001. Epub 2012 Aug 31. PMCID: PMC3432069 [Open Access]
- Enderling H, Hahnfeldt P, Hlatky L, Almog N. Systems biology of tumor dormancy: linking biology and mathematics on multiple scales to improve cancer therapy. Cancer Res. 2012 May 1;72(9): 2172-5. Epub 2012 Mar 13. PMCID: PMC3374524 [Open Access]
- Almog N. Molecular mechanisms underlying tumor dormancy. Cancer Lett. 2010 Aug 28;294(2):139-46. Epub 2010 Apr 2.
- Almog N, Klement GL. Platelet proteome and tumor dormancy: can platelets
content serve as predictive biomarkers for exit of tumors from dormancy? Cancers (Basel). 2010 May 11;2(2):842-58. PMCID: PMC3835108 [Open Access]
- Fainaru O, Almog N, Yung CW, Nakai K, Montoya-Zavala M, Abdollahi A, D'Amato R, Ingber DE. Tumor growth and angiogenesis are dependent on the presence of
immature dendritic cells. FASEB J. 2010 May;24(5):1411-8. Epub 2009 Dec 14. PMCID: PMC2879945 [Open Access]
- Enderling H, Anderson AR, Chaplain MA, Beheshti A, Hlatky L, Hahnfeldt P. Paradoxical dependencies of tumor dormancy and progression on basic cell kinetics. Cancer Res. 2009 Nov 15;69(22):8814-21. Epub 2009 Nov 3. [Open Access]
- Fakir H, Tan WY, Hlatky L, Hahnfeldt P, Sachs RK. Stochastic population dynamic effects for lung cancer progression. Radiat Res. 2009 Sep;172(3):383-93.
- Almog N, Ma L, Raychowdhury R, Schwager C, Erber R, Short S, Hlatky L, Vajkoczy P, Huber PE, Folkman J, Abdollahi A. Transcriptional switch of dormant tumors to fast-growing angiogenic phenotype. Cancer Res. 2009 Feb 1;69(3):836-44. Epub 2009 Jan 27. [Open Access] COVER ARTICLE
- Cervi D, Yip TT, Bhattacharya N, Podust VN, Peterson J, Abou-Slaybi A, Naumov GN, Bender E, Almog N, Italiano JE Jr, Folkman J, Klement GL. Platelet-associated PF-4 as a biomarker of early tumor growth. Blood. 2008 Feb 1;111(3):1201-7. Epub 2007 Oct 3. [Open Access]
- Almog N, Henke V, Flores L, Hlatky L, Kung AL, Wright RD, Berger R, Hutchinson L, Naumov GN, Bender E, Akslen LA, Achilles EG, Folkman J. Prolonged dormancy of human liposarcoma is associated with impaired tumor angiogenesis. FASEB J. 2006 May;20(7):947-9. Epub 2006 Apr 25. [Open Access]
- Sachs RK, Hlatky LR, Hahnfeldt P. Simple ODE models of tumor growth and anti-angiogenic or radiation treatment. Math Comput Model. 2001 Jun;33(12-13):1297-305. Epub 2001 May 24. [Open Access]
- Hahnfeldt P, Panigrahy D, Folkman J, Hlatky L. Tumor development under angiogenic signaling: a dynamical theory of tumor growth, treatment response, and postvascular dormancy. Cancer Res. 1999 Oct;59(19):4770-5. [Open Access]